It is said that through gold plating, your metal piece can be more durable and can resist corrosion that is brought by the moisture in the environment. In some other cases, re-plating is done so that the old metal piece does not need to be thrown away since it can be reverted to its original condition. However, have you ever wondered why how it is possible? Is there any science behind such technique? There is another way to gold plating. It is more reliable and the process is clear. It is called electroplating. What is electroplating? Is it needed? Or it is just a way to trick buyers to think that it is better than other gold plating solutions? You judge it based on the explanation that can be found in this infographic.
1. Electroplating improves the electrical conductivity of a base material (e.g. gold). It also enhances the ability of the material to resist corrosion so that even if it will be exposed to a high level of moisture, it would only give minimal effect or zero effect on the metal piece. Another advantage of electroplating is its solderability or a capacity of the material to be united with another material. Such could be enough to justify why you need to opt for electroplating.
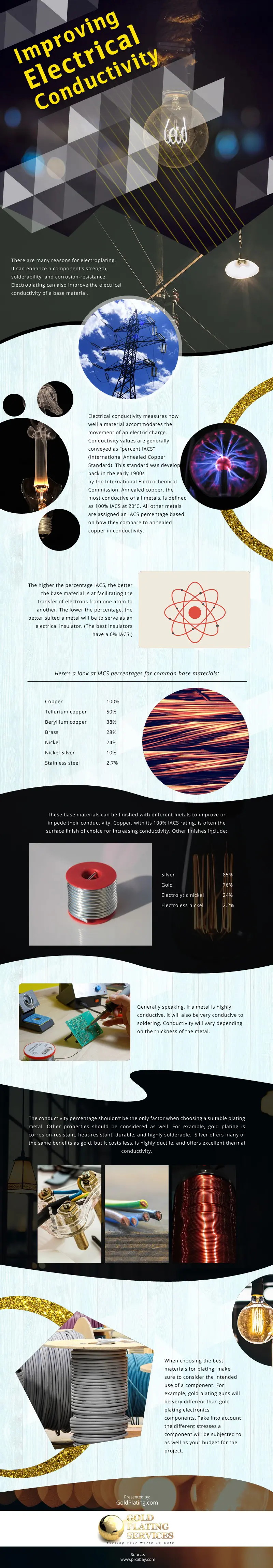
2. However, for you to appreciate electroplating, you need to understand what is electrical conductivity. It is a term that gauges how efficiently does a material accommodate or channels the flow of electric charge. The level of conductivity is measured in terms of “percent IACS” where IACS stands for International Annealed Copper Standard. The most conductive metal is Annealed copper that is why its electrical conductivity becomes the basis for measuring the conductivity of other materials. Actually, such measurement has been developed since the 1900s by the International Electrochemical Commission which makes it a reliable measuring technique.
3. The best conductors have percent IAS because it means that the transfer of electrons of an atom of a base material can be easily transferred or flown to another atom. However, the lower the percent IACS means that the conductivity is low which you can translate to insulation. If it has high percent IACS, it is a conductor. However, if the percent IACS is low, the material is an insulator.
4. Since annealed copper is the basis of other materials for measuring conductivity, it is given that it has a 100% IACS. It is followed by tellurium copper at 50%, beryllium copper with 38%, Brass with 28%, nickel with 24%, nickel silver with 10% IACS and stainless steel of 2.7% IACS. This means that if you are planning to pursue with electroplating, it is better than you use copper and not stainless steel.
5. However, in spite of this fact, it is still advised that you don’t solely depend on conductivity percentage. This is because you might be able to find an alternative that is less costly. Gold plating could be a good option because it provides durable and corrosion resistant finish yet if you opt for silver almost the same properties can be achieved but at a lower price.
source: https://www.goldplating.com/blogs/news/improving-electrical-conductivity
Comments
Download this infographic.






